Across hospitals, clinics, and diagnostic labs worldwide, thousands of medical devices still operate electronics designed decades ago. These systems continue to perform essential clinical functions, yet their internal architectures often rely on obsolete components, undocumented firmware behavior, and manufacturing processes that are increasingly difficult to sustain.
Legacy devices are rarely replaced quickly in healthcare environments. The cost of replacement, clinical validation, and regulatory approval makes modernization more complex than simply building a new product. But that complexity also presents an opportunity.
When approached strategically, redesigning legacy medical electronics can deliver measurable benefits: improved performance, stronger regulatory alignment, lower maintenance costs, and extended product lifecycles.
Through years of Medical Device Hardware Design and Hardware Firmware Development, we’ve seen how legacy redesign projects can transform aging systems into future-ready medical platforms.
What follows is the structured methodology we use to modernize legacy medical devices safely and effectively.
Why Legacy Medical Devices Still Matter
Healthcare technology evolves quickly, but clinical infrastructure does not always keep pace. Many hospitals rely on reliable but outdated devices.
Common challenges with legacy medical electronics include:
- Obsolete microcontrollers and analog components
- Limited firmware update capability
- Incomplete or missing documentation
- Difficulty sourcing replacement parts
- Growing regulatory compliance gaps
Despite these limitations, the core clinical functionality of these devices often remains valuable. Replacing them entirely may be unnecessary and sometimes impractical.
This is where structured redesign becomes powerful. Legacy systems contain years of clinical insight embedded in their design. The goal is not to discard that knowledge, but to modernize it through disciplined engineering.
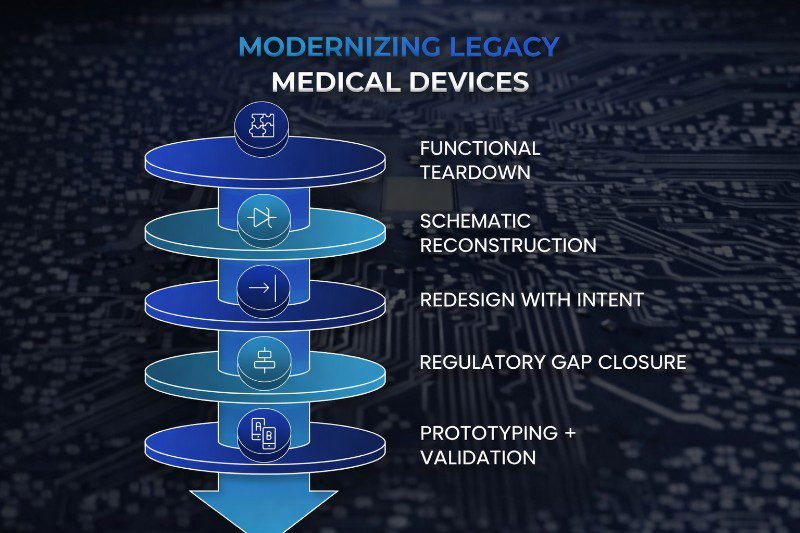
Step 1: Functional Teardown and Behavioral Mapping
The first step in legacy redesign is understanding what the device truly does, not just what documentation claims it does.
We begin with a functional teardown to identify the primary subsystems:
- Power architecture
- Control logic
- Sensing circuitry
- User interface components
- Communication interfaces
Because legacy documentation is often incomplete, we rely heavily on:
- Signal tracing
- Oscilloscope measurements
- Logic analyzers
- Runtime observation
This phase reveals undocumented behaviors that must be preserved during redesign. In medical devices, small timing interactions or analog signal characteristics can be clinically significant.
Functional teardown ensures that modernization does not unintentionally alter validated device behavior.
Step 2: Schematic Reconstruction
Once a functional understanding is established, the next step is to reconstruct the electronic design.
We reverse engineer PCB netlists from:
- Board scans
- Component identification
- Continuity testing
- CAD recreation
This allows us to rebuild schematics that accurately reflect the device’s current configuration.
During this process, we identify:
- Obsolete components
- Single-source dependencies
- Thermal risks
- Power inefficiencies
- Layout limitations
This stage forms the foundation for modern Medical Device Hardware Design, enabling redesign decisions to be made with confidence.
Step 3: Redesign With Intent
Redesigning a legacy medical device is not about replacing parts one-for-one. It is about improving reliability, maintainability, and future scalability while preserving clinical behavior.
Typical redesign improvements include:
- Replacing legacy microcontrollers with low-power secure SoCs
- Improving power management architecture
- Modularizing system components
- Preparing for wireless connectivity such as BLE or LTE-M
- Enabling firmware update capability
This is where Hardware Firmware Development becomes central. Hardware decisions must anticipate future firmware requirements, and firmware architecture must leverage new hardware capabilities.
The redesigned system should remain clinically familiar while becoming technologically adaptable.
Step 4: Closing Regulatory Gaps
Legacy devices often predate modern regulatory expectations. Updating electronics without addressing compliance gaps can create future approval challenges.
We align redesign efforts with:
- IEC 60601 safety requirements
- ISO 13485 quality management expectations
- MDR documentation needs
- Serviceability and traceability requirements
Engineering improvements may include:
- Adding diagnostic interfaces
- Integrating self-test routines
- Improving logging capabilities
- Enhancing electrical safety margins
- Designing for auditability
Regulatory alignment is not an afterthought; it is integrated into the redesign process from the beginning.
This reduces future submission risk and simplifies lifecycle management.
Step 5: Firmware Integration and Validation
Modernizing hardware requires equally thoughtful firmware integration.
Legacy firmware is often:
- Undocumented
- Tightly coupled to hardware
- Difficult to update
- Inefficient in memory or power usage
Through structured Firmware Development Services, we rebuild firmware architecture to support:
- Modular functionality
- Predictable timing behavior
- Maintainable code structure
- Upgrade readiness
- Improved diagnostics
Validation then compares:
- Original device behavior
- Redesigned system performance
- Edge-case scenarios
- Long-duration operation
The goal is not only to match legacy behavior, but to enhance reliability while preserving clinical confidence.
Extending Product Life Through Redesign
One of the greatest benefits of legacy redesign is lifecycle extension.
Rather than forcing healthcare providers to replace trusted systems, modernization allows manufacturers to:
- Maintain product continuity
- Reduce manufacturing risk
- Improve serviceability
- Support future firmware enhancements
- Strengthen regulatory positioning
Legacy hardware becomes a platform for innovation instead of an obstacle.
Engineering the Future from the Past
Legacy medical devices represent years of clinical validation, user familiarity, and proven workflows. Redesigning them responsibly requires both technical precision and respect for their operational history.
When executed correctly, redesign projects deliver:
- Improved performance
- Reduced maintenance costs
- Better compliance alignment
- Extended product lifespans
- Stronger patient safety margins
Legacy systems are not outdated failures; they are engineering foundations waiting to be modernized.
Final Thoughts
Modernizing legacy medical electronics requires more than component replacement. It demands a structured approach that balances preservation, innovation, and regulatory responsibility.
At Pinetics, we specialize in Medical Device Hardware Design, Firmware Development Services, and Hardware Firmware Development to help manufacturers transform legacy systems into future-ready medical platforms. By combining reverse engineering expertise with modern embedded design practices, we enable healthcare technologies to evolve without losing their proven clinical value.
Legacy isn’t a dead end. It’s a blueprint for smarter reinvention engineered carefully for tomorrow’s healthcare systems.
 Sr. Test Engineer
Sr. Test Engineer Sales Marketing Manager
Sales Marketing Manager Marketing & Sales – BBA : Fresher
Marketing & Sales – BBA : Fresher