In most industries, firmware is judged by whether it works. In MedTech, firmware is judged by whether it can be proven to work safely and consistently under every foreseeable condition.
You cannot patch your way out of non-compliance. And you cannot debug your way past a failed audit. In medical devices, firmware is not just logic running on silicon. It is liability, evidence, and accountability all compiled into one binary.
Yet one of the most common failure patterns we see in medical device programs is this:
Highly skilled firmware engineers build elegant systems without accounting for regulatory context, only to be forced to undo months of work.
Not because the firmware was wrong. But because it was undocumented, untraceable, or misaligned with regulatory expectations.
The Cost of Ignoring Regulatory Context
Firmware engineers are trained to think in terms of:
- Performance
- Memory efficiency
- Timing determinism
- Fault tolerance
Regulators, however, think in terms of:
- Risk classification
- Traceability
- Repeatability
- Documented intent
When these worlds don’t meet early, the consequences are severe:
- Partial rewrites of core firmware modules
- Redesign of state machines and safety logic
- Re-validation of test suites
- Delayed submissions and missed market windows
This is not theoretical. It happens constantly across MedTech programs.
In Medical Device Hardware Design and firmware co-development, factoring regulatory demands into design is essential. Compliance is a fundamental constraint, just as much as power or memory, and it impacts product viability.
Firmware Is Regulated Software, Not Just Embedded Code
Medical device firmware does not exist in isolation. It operates as part of a regulated system governed by standards such as:
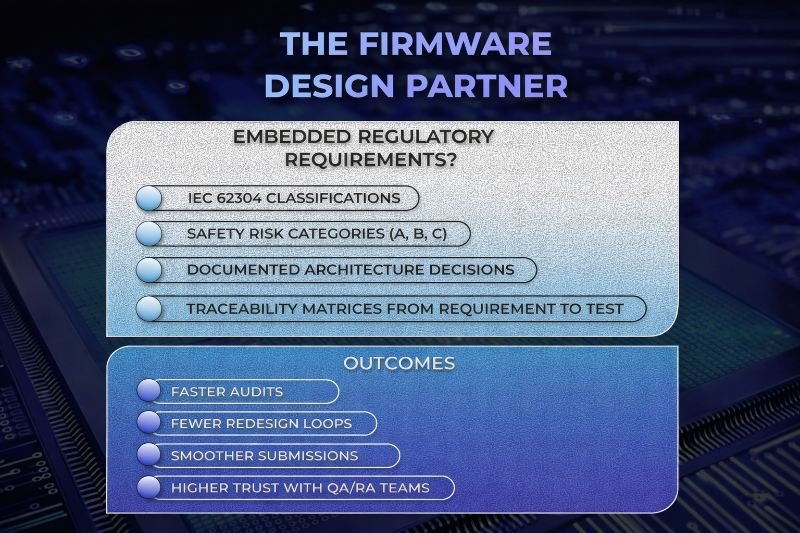
- IEC 62304 (medical software lifecycle)
- ISO 14971 (risk management)
- IEC 60601 (safety and performance)
Under IEC 62304, software is classified by risk level:
- Class A – no injury possible
- Class B – non-serious injury possible
- Class C – serious injury or death possible
This classification directly affects:
- Architectural requirements
- Verification depth
- Documentation rigor
- Change management expectations
If firmware engineers misunderstand, overlook, or assume the finalized safety class late in development, design choices often will not meet regulatory expectations and become non-compliant.
The regulatory context does not sit above firmware. It prescribes the required methods, documentation, and considerations for structuring and validating firmware to meet regulatory requirements.
Traceability Is a Firmware Design Requirement
One of the most misunderstood regulatory concepts among engineers is traceability.
Traceability is not a spreadsheet maintained by QA after the fact. It is a design philosophy.
Every regulatory reviewer expects a clear chain:
- User need → System Requirement → Software Requirement → Implementation → Verification → Validation
If a firmware engineer writes code without mapping it to an explicit requirement, that code becomes a liability. During an audit, undocumented logic is treated as uncontrolled behavior under regulatory standards, even if it works perfectly.
In robust Firmware Development Services, traceability is embedded into:
- Function definitions
- Commit messages
- Test cases
- Design documentation
Code must be defensible, not just functional.
Architecture Decisions Must Be Justified, Not Assumed
In consumer electronics, architectural decisions are often evaluated pragmatically: “Does it work?” “Is it efficient?”
In MedTech, the question becomes: “Why was this architecture chosen, and what risks does it mitigate?”
Firmware architecture must be documented with:
- Justification for task separation
- Rationale for scheduling models
- Explanation of fault handling behavior
- Evidence of safe failure modes
This is where Hardware Firmware Development intersects directly with regulatory scrutiny.
For example:
- Why is a watchdog reset acceptable here?
- Why is this interrupt prioritized over another?
- What happens if this peripheral fails silently?
If those answers are not documented, regulators assume they were never considered.
Risk Management Is Not a QA Exercise
Treating risk management as an isolated activity for QA or RA teams ignores regulatory mandates. Firmware engineers must integrate risk controls in core design, as required by compliance standards. Firmware engineers implement risk controls.
If a hazard analysis identifies a risk of incorrect output, the mitigation may be:
- Redundant computation
- Sanity checking
- Timeout enforcement
- Safe state transitions
Those mitigations live in firmware. If firmware engineers are not fluent in risk language, mitigations become implicit rather than explicit, making them impossible to properly validate.
In Medical Device Hardware Design, risk flows downward into firmware behavior. That flow must be intentional.
Regulatory Thinking Changes How Firmware Is Written
When firmware engineers speak regulatory language, several things change immediately:
1) Code Becomes Intentional: Logic is written with an explicit purpose tied to requirements, not convenience.
2) Edge Cases Are Designed, Not Discovered: Failure modes are anticipated, documented, and tested, but not found during audits.
3) Testing Becomes Structured: Verification is executed to demonstrate compliance with the specific regulatory safety class, going beyond simple functional testing to satisfy authorities.
4) Documentation Is Continuous: Design decisions are recorded while context is fresh, not reconstructed months later.
This discipline dramatically reduces late-stage rework.
Faster Approvals Come from Better Engineering, Not Better Paperwork
A common misconception is that regulatory speed comes from better documentation teams. Speed comes from regulatory-aware engineering.
When firmware is built correctly:
- Audits move faster
- Reviewer questions decrease
- Non-conformities are rare
- Submission cycles shorten
Engineering teams that treat regulatory language as a core skill consistently outperform those that treat it as external bureaucracy.
Firmware, Hardware, and Regulation Are Interdependent
In MedTech, firmware does not exist without hardware, nor without regulation.
For example:
- Hardware safety features must be supported by firmware behavior.
- Firmware fault handling must match hardware failure modes.
- Power management decisions must align with safety expectations.
This is why Hardware Firmware Development must be coordinated from the earliest design phases.
Regulators evaluate the device as an integrated system, requiring full compliance and harmonious behavior across firmware and hardware to meet approval standards.
The Real Risk of “Fixing It Later”
Perhaps the most dangerous mindset in medical device firmware development is:
“We’ll make it compliant later.”
Once firmware architecture is locked, changing it to meet regulatory expectations often requires substantial rework, making compliance expensive, not because regulations are unreasonable, but because engineering choices were made without regulatory awareness.
Once firmware architecture is locked, changing it to meet regulatory expectations often requires:
- Refactoring task models
- Rewriting error handling
- Redesigning state machines
- Re-executing verification plans
At that point, compliance becomes expensive not because regulations are unreasonable, but because engineering choices were made without regulatory awareness.
Compliance as a Design Partner, not a Constraint
The most successful MedTech teams do not fight regulations. They collaborate with it.
When firmware engineers speak regulatory language:
- QA and RA teams become collaborators, not blockers.
- Design reviews become clearer and faster.
- Audits feel predictable instead of adversarial.
Regulation stops being something to survive and starts becoming something to leverage.
Final Thoughts
In MedTech, firmware engineers do not just write code. They write evidence. Every task scheduler, interrupt handler, and state transition must stand up not only to runtime conditions but to regulatory scrutiny.
At Pinetics, we approach Medical Device Hardware Design, Firmware Development Services, and Hardware Firmware Development with this reality at the core. We help engineering teams build firmware that is not only efficient and robust, but also traceable, auditable, and regulator-ready from the first line of code.
Success in medical technology isn’t about a device working once. It is defined by whether it can be proven to work safely, repeatedly, and under all conditions that regulators expect.
When firmware engineers speak the language of regulation, innovation accelerates rather than stalls. That is how prototypes become trusted clinical products.
 Sr. Test Engineer
Sr. Test Engineer Sales Marketing Manager
Sales Marketing Manager Marketing & Sales – BBA : Fresher
Marketing & Sales – BBA : Fresher